  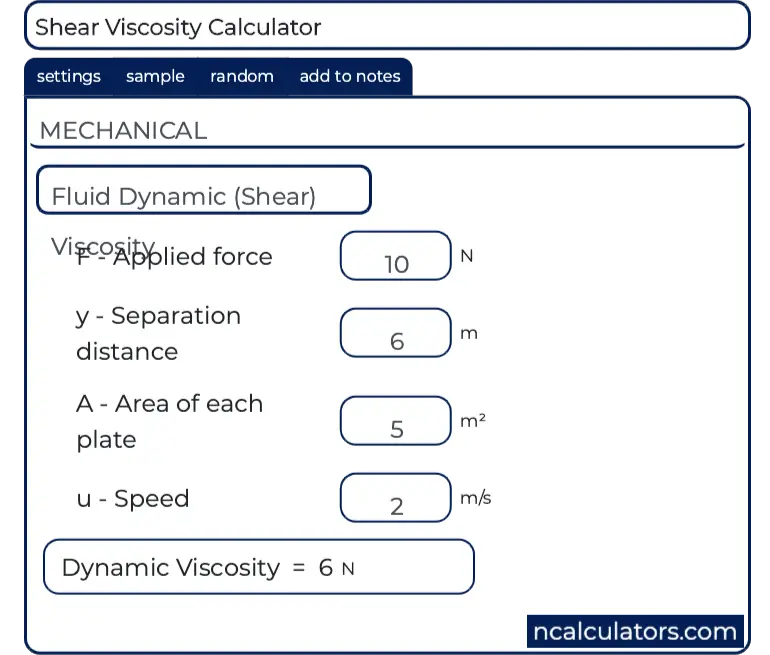 
If the set of conditions entered in the model generate negative answers, then clearly it is necessary to adjust the variables as appropriate until a realistic result is obtained. With this being the case, they will be accurate to within 10% of the actual results obtained. The graphs above are generated from calculations assuming the hose to be in good condition and laid in a straight line. If the head is given in metres of water, each 1-metre head (3.28 ft) induces 0.1 bar (1.47 psi.) pressure.Īll formulae for finding the amount of fluid that will flow through a hose at a given time, are approximate. The pressure is sometimes stated as ‘head of water’. The surface character of the bore, the number, and shape of bends incorporated in the run of the hose also influence the flow rate. The quantity of fluid that will be discharged through a hose depends on the pressure applied at the feed end, the hose length and bore diameter. Kindly refrain from reaching out to us with any inquiries related to the utilization of these charts. These charts are intended for guidance purposes only. ĭensity of crude oil, Density of fuel oils, Density of lubricating oil and Density of jet fuel as function of temperature.Īs shown in the figures, the change in density is not linear with temperature - this means that the volumetric expansion coefficient for water is not constant over the temperature range.Quantity Fluid Flow vs Bore Diameter Data 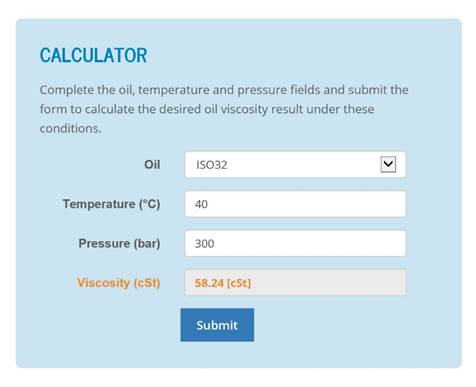
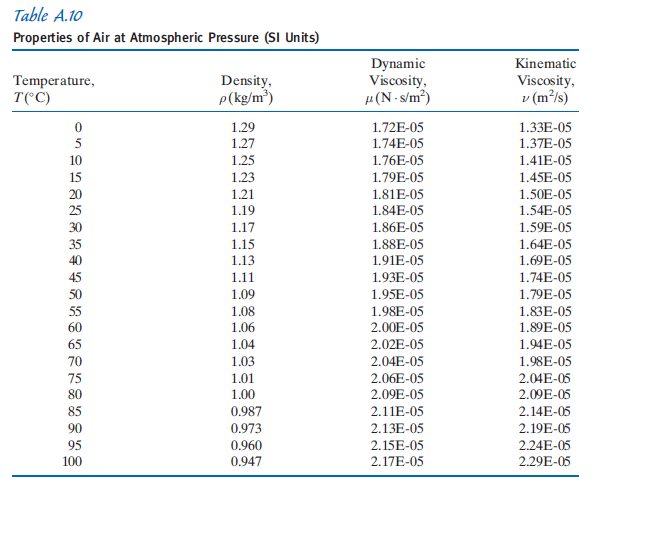
See also other properties of Water at varying temperature and pressure: Boiling points at high pressure, Boiling points at vacuum pressure, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pK w, of normal and heavy water, Melting points at high pressure, Prandtl number, Properties at Gas-Liquid Equilibrium Conditions, Saturation pressure, Specific gravity, Specific heat (heat capacity), Specific volume, Thermal conductivity, Thermal diffusivity and Vapour pressure at gas-liquid equilibrium.įor other substances, see density and specific weight o f acetone, air, ammonia, argon, benzene, butane, carbon dioxide, carbon monoxide, ethane, ethanol, ethylene, helium, hydrogen, methane, methanol, nitrogen, oxygen, pentane, propane and toluene. See Water and Heavy Water for thermodynamic properties at standard condtions. The density of water depends on temperature and pressure as shown below: Note! Temperature must be within the ranges 0-370 ☌, 32-700 ☏, 273-645 K and 492-1160 °R to get valid values. The output density is given as g/cm 3, kg/m 3, lb/ft 3, lb/gal(US liq) and sl/ft 3. The calculator below can be used to calculate the liquid water density at given temperatures. See more about the difference between mass and weight Online Water density Calculator The density of water is 1.940 sl/ft 3 at 39 ☏ (4 ☌), and the specific weight in Imperial units is In the Imperial system the mass unit is the slug, and is derived from the pound-force by defining it as the mass that will accelerate at 1 foot per square second when a 1 pound-force acts upon it:ġ = 1 * 1 and 1 = 1 /1 In the SI system, specific weight of water at 4☌ will be: G = acceleration due to gravity, units typically and value on Earth usually given as 9.80665 m/s 2 or 32.17405 ft/s 2 Specific weight is the ratio of the weight to the volume of a substance: Pure water has its highest density 1000 kg/m 3 or 1.940 slug/ft 3 at temperature 4☌ (=39.2☏). Density is the ratio of the mass to the volume of a substance: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
